Additional information
1013621-79-8
ACI-017601
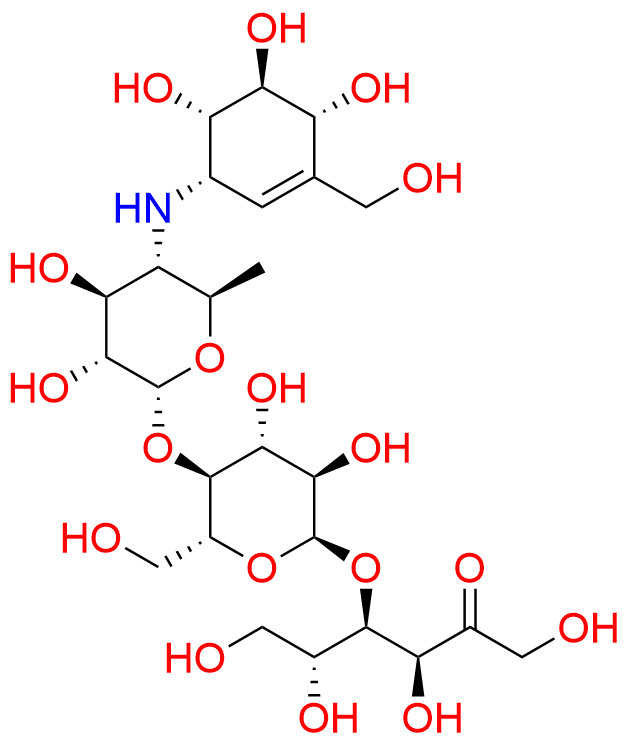
O-4,6-dideoxy-4-[[(1S,4R,5S,6S)-4,5,6-trihydroxy-3-(hydroxymethyl)cyclohex-2-enyl]amino]-?-D-glucopyranosyl-(1?4)-O-?-D-glucopyranosyl-(1?4)-D-arabino-hex-2-ulopyranose
C25H43NO18
430.37
White Powder
NA
O-4,6-Dideoxy-4-[[(1S,4R,5S,6S)-4,5,6-trihydroxy-3-(hydroxymethyl)-2-cyclohexen-1-yl]amino]-a-D-glucopyranosyl-(1?4)-O-a-D-glucopyranosyl-(1?4)-D-fructose; Acarbose EP Impurity A
Acarbose D-Fructose Impurity
Antidiabetic
Pharmaceutical Impurity Standards
![N-ETHYL AZILSARTAN MEDOXOMIL (25 mg) ((5-Methyl-2-oxo-1,3-dioxol-4-yl)methyl 2-ethoxy-1-{[2′-(4-ethyl-5-oxo-4,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-1H-benzimidazole-7-carboxylate)](https://analyticachemie.in/wp-content/uploads/uspimage-1.jpg)

